15. ‘Dicalcic’ vs Reactive Phosphate Rock (RPR), elemental S and Fine Lime (updated September 2019)
What exactly are you getting for your money?
Dicalcic Super
Dicalcic (aka lime-reverted superphosphate) has been around a long time. When properly made, the water-soluble P component in super, called monocalcium phosphate or MCP, is fully converted after crushing by chemical reaction with lime, to a different form of P called dicalcium phosphate or DCP (hence the name ‘dicalcic’).
A well made straight superphosphate should contain at least 90-95% of its total P in the form of water-soluble MCP. The difference between this – say 8.0% P and the total P content of say 9.0%, is comprised of agronomically very ineffective, unreacted manufacturing phosphate rock and various complex iron-aluminium-calcium phosphates of very dubious value to plants. Interestingly, since the demise of the Fertilisers Act, superphosphate manufacturers now clearly imply, in the way they and their field staff use the total P rather than the water-soluble P content of super to calculate how much super to apply per hectare, that this non water-soluble P component is all plant-available.
Chemically, it should only be necessary to thoroughly mix in 30-35% of lime by weight to achieve full conversion of the MCP to DCP over a couple of days. For a variety of reasons, the main one I think being reducing the amount of mixing effort required, much higher proportions of lime – typically 50% – are used. This means that the total P of the product has been diluted from 9.0% P to only 4.5%, and what was water-soluble MCP component of say 8.0% P, to a mere 4.0% P of DCP.
It is extremely important to ask and try to answer the question “is it worthwhile for the farmer to use dicalcic and why?”
Manufacturers and suppliers claim that (1) the DCP form of P is a far more efficient per unit P than is water-soluble MCP, and (2) the lime content helps the plant to utilise all soil nutrients. DCP is not water soluble, but unlike the very unavailable forms of non water-soluble P in straight super, is very easily converted to plant-available water-soluble P in the soil. Probably the main specific advantage of applying P as DCP instead of MCP is that, by being initially non water-soluble, it avoids much of the severe run-off risk that the water-soluble P in straight super faces, when a rainfall or irrigation-induced surface water run-off event in the weeks after application can easily result in a kg or more per hectare of P ending up in the nearest waterway.
This is the direct cause of much of the increasing eutrophication of our lakes and rivers – nitrate leaching from urea and cow urine patches being the other major contributors.
As deeply serious as this is environmentally, it typically represents less than 10% of the P that is applied. So how does this fit with the claims that dicalcic super only needs to be applied at the same rate per hectare as straight super – in other words, at only half the rate of P, clearly implying that it is somehow twice as effective per unit P as straight super.
Rather than totally rubbish this claim, as some of my ex fellow scientists have done and continue to do, we need to dig a bit deeper, if you will excuse the pun. In my experience, farmer observation, despite the fact that it may not be based on the results of scientifically-conducted, fully replicated and statistically analysed trials, are not to be ignored. Time is a great leveler. I believe it is no coincidence that the deepest farmer support for dicalcic super is in areas of yellow grey and yellow brown soils, particularly where there is ready access to cheap lime, and family continuity in farm ownership, meaning long-term observation of cause and effect, and the passing on of these observations.
It so happens that the yellow grey earth soils in particular are known to be (a) capable of accumulating huge amounts of P in very inert organic form, and (b) capable of having this organic P mobilised if lime is applied. I observed this for myself on the ultra-long term superphosphate trial at the MAF’s Winchmore Irrigation Research Station where I was stationed from 1974-82. When the whole trial was limed for the first time for 20 years in 1975, there was a massive mobilisation of soil organic P into plant available form, as measured by Olsen P soil tests and plant uptake.
The question within the question then, is whether applying dicalcic super annually is any more effective than applying straight super annually, and is there a need for a heavy rate of lime separately every 5 years or so, even on these soils? Most agricultural scientists would say no. I would say probably yes, with some provisos, and certainly not to the extent of being able to halve the rate of application of P long term. My estimate is that a 25-30% reduction is possible.
The remainder of the benefit in improved efficiency that cannot be explained by the 10% improvement resulting from reduced P run-off comes, I believe, from the intimate contact between P – now, remember, in the alkaline DCP form – and lime in dicalcic super provides a stimulation of soil microbiological activity, resulting in improved turnover of all nutrients held largely in organic form, meaning not just P, but N and S and trace elements as well. Faster turnover of nutrients provides more opportunity for root growth and nutrient uptake.
Other major soils, especially the allophonic ash soils whose depth and drainage, combined with reliable rainfall, have made them the mainstay of dairy farming in this country, store proportionately far more of their P in inorganic form fixed to clay particles. They have a naturally higher rate of biological activity and this, combined with a natural high pH buffering capacity, means that there is not the same extent of P-release benefit from lime. The real challenge on the ash soils is finding out how to prevent the P being fixed by allophone clay before the plant can use it. But that’s another story.
RPR, S and (reasonably) fine lime
So, I’ve said that dicalcic is possibly 25-30% more efficient per unit P than straight super and lime applied separately on the yellow grey earth and possibly yellow brown earth soils, but not much elsewhere. It is also better for the environment because of reduced run-off. But it is about 40-50% more expensive on an applied basis. So is there a better way? I believe there is – blends of RPR, S and reasonably fine lime. Let me explain why.
If we go back to dicalcic for a minute, it is made by reacting straight super with lime. Manufacturers of single superphosphate (SSP) have gone to the trouble of acidulating a non-recative phosphate rock with concentrated sulphuric acid, to convert the phosphate into a plant-available form, and then semi-granulating it so it can be handled more easily. This costs money, and the product has no liming value. So where is the logic in taking that, crushing it up again, mixing it with lime to change the type of phosphate into a non water-soluble form again, all to give the final product a liming equivalent of 50% of the same weight of lime? No wonder it costs 40% more per unit P than SSP!
So all in all, an expensive process, which also results in high transport and spreading costs per unit P because of the low P content. Lets say you want to put on 15 kg of available P per hectare, typically enough to maintain 10 SU/ha. At 4.0% P available P in dicalcic, that means 375 kg/ha is required. This will also provide the liming equivalent of about 135 kg/ha of lime – the typical annual requirement on a reasonably high-producing non-ash soil. However, the reality is that few users of dicalcic put on this amount, because of the high transport and spreading costs, and therefore soil P levels continue to slowly decline, while soil pH tends to be more or less maintained.
Lets look now at RPR and lime. RPR, or reactive phosphate rock, typically contains 12.5-13% P. (Warning 2015: recently, some suppliers, including at least one of the large companies, have been importing completely unbeneficiated RPR ore from Peru containing about 40% inert clay, reducing the P content to 7-8%. Often, it is claimed to be much higher, such as 11.5%P. To do this is theft. Sometimes, it is blended with a non-RPR to increase the overall citric solubility. This is also theft. If you are in any doubt whatsoever about the P content of what you are buying, tell the supplier you well be taking a representative sample from the truck when it arrives at the farm, and getting it analysed at an independent laboratory such as Hills).
The definition of an RPR as used in New Zealand is a natural phosphate rock which, because of the way it was formed, will dissolve fast enough per year to supply sufficient P to maintain the growth of a high-producing pasture at soil pH levels of up to 6.0, which is all you need.
RPR automatically contains some of its high calcium content in carbonate form, which is what lime is, and it gives each tonne of RPR the liming ability of 0.5 tonne of high-quality lime. This is actually what makes RPR more ‘reactive’ – meaning more quickly released into plant-available form in the soil – than ordinary phosphate rock, which has to be acidulated with sulphuric or other acids. It also consumes soil acidity during this process.
So, lets say you wanted to put on 15 kg P/ha as RPR (120 kg RPR/ha). Add in 10 kg/ha of fine elemental S, which is all you need because it doesn’t leach unlike the sulphate-S in super and dicalcic, and you still have a net liming effect of about 30 kg/ha. To maintain soil pH in the 5.6-5.7 range typically requires an application of 150 kg lime/ha annually. This 150 minus the 30 from the RPR/S leaves 120 kg/ha to be added in as lime, giving a total application of 240 kg/ha of this 50/50 mixture.
Note that the lime does not need to be expensive $200/tonne superfine minus 20 micron lime. Minus 100 microns is fine enough, and doesn’t cost the earth (typically $70-80/t). To that the components of the mix (RPR, lime and elemental S) are all very evenly distributed as they land on the ground, we need to apply the mix in a slightly damp form. A dry mix will be subject to segregation and drift.
A damp mix may not discharge evenly from the truck or aircraft, and in fact can be very dangerous in the latter case. A high water-content suspension will be very expensive to apply because of the cost of transporting and spreading the weight of water. In addition, the likelihood of settling out of high water-content, so-called ‘suspensions’ in aircraft tanks means application rates can be variable.
A high-solids fluid – typically only 15-25% water – actually stays far more uniform, because the water content is far more tightly held. Further, each droplet as it lands contains the same proportion of the components. But the cost of making the fluids, keeping them mixed in the aircraft tank during application is just too high (see 2011 costings).
The simple solution is to spray the dry mix with about 5% water as it leaves the aircraft. Instead of more than doubling the spreading cost as with suspensions and fluids, it adds a mere 5-10%.
Dicalcic Super vs Wetted RPR/S/Fine Lime – Cost Comparison Example
Objective – to apply hill country maintenance P (15 kg/ha) and S (12 kg/ha as sulphate or 10 kg/ha as fine elemental S, plus sufficient lime, in fine enough form, to stimulate soil microbial activity and maintain soil pH.
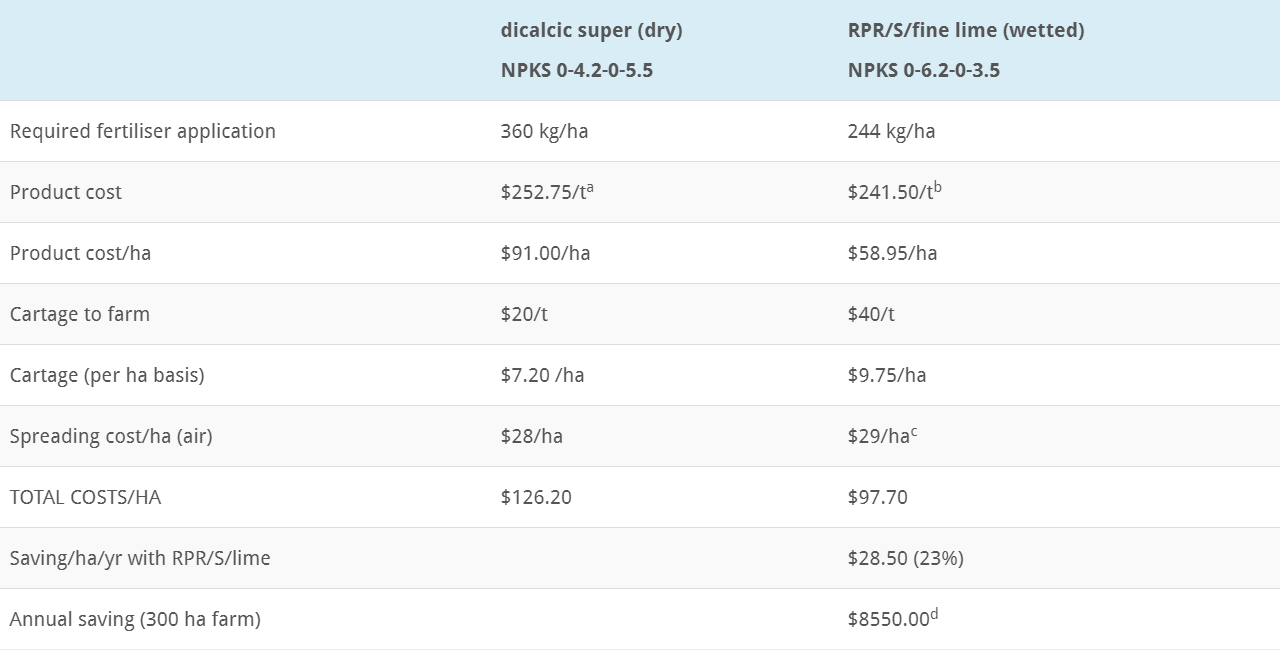
Notes: prices ex Ballance price list
(a) ex Wairoa
(b) ex Hastings, assuming $383 for RPR/S and $100/t for finer lime and blending – 50/50 blend gives $241.50/t
(c) $4/ha lower rate for lower fert weight/ha, plus 10% for water spray
(d) the $8550 will pay for a trip to Disneyland for the family, or a new quad This demonstrates the large savings in maintaining hill-country P, S and liming.
Lime (and the calcium it brings with it) should be treated no differently than the fertiliser nutrients. That is, we should be striving to keep pH levels as near to the optimum hill country of 5.6-5.7 continuously, rather than suffering a widely varying soil pH during a 5 year cycle.
Final Comments: Other options such as fixed-wing application of annual superphosphate plus capital lime (say 1 tonne/ha every 5 years), or RPR/S plus reduced capital lime are typically slightly cheaper averaged over 5 years. However, what often happens is that, if cash-flow is not good when the capital lime is due, the pH can fall to a level where aluminium toxicity becomes a real problem, reducing pasture growth and N fixation by clover, which in turn will have a negative effect on soil biological activity.
BUYER BEWARE Farmers must be alert to the existence of unscrupulous ‘muck and mystery’ merchants who use the mask of attractively sign-written helicopters and very slick marketing to promote the use of very costly low-nutrient dilute sprays which are claimed to contain components that will free up nutrients locked up in the soil. It would be good if these components could be shown to achieve something, and unfortunately this has not been the case. By the time you realise this, in 2 or 3 years, your production will have fallen 10%. Sad but true.
